Compound Azintamide Enteric-coated Tablets, Pancreatin supplement with simethicone, effectively promote choleretic, quickly eliminate abdominal distension, effectively relieve indigestion symptoms such as loss of appetite, abdominal discomfort, early satiety, belching, fatty stool, chemical indigestion, etc.

CSL Behring is one of the largest import suppliers in China's human albumin market. Baheal has collaborated with CSL Behring since 2018 and remains a key partner for CSL Behring in China. Human albumin is a life-saving product primarily used to treat shock caused by hemorrhagic trauma or burns, as well as for the prevention and treatment of hypoalbuminemia. Due to factors such as stringent market regulation, increased clinical demand, and limited plasma supply, China's human albumin market has consistently faced a supply shortage.

Micro-needle eye mask, applying the "transdermal drug delivery technology” originated from a key invention of a Japanese medical laboratory, enabling users to have medical-level care at home. Medical evidence shows safe and effective release of worries about trauma, pain, escharotic and reconstruction. With simple ingredients, high efficacy and 40+ patents, it effectively relieves the symptoms of dark circles, fine lines around the eyes, etc.

Bi Bai Ke originates from the Qing Dynasty imperial prescription “Wan Ying Ding,” a formula for clearing heat and detoxifying the body. It uses natural bear bile powder as its primary ingredient, with each tablet containing 11.1mg of bear bile powder. It is formulated with authentic herbs such as wine-processed rhubarb and Coptis chinensis, following a rigorous and well-balanced formulation. It is specifically used for various symptoms of “heat” in the body, offering unique therapeutic effects.

Enrolled in China National Reimbursement Drug List (NRDL) category B list, level I medical evidence. First anti-hepatic fibrosis therapeutic drug recommended in six Major Guidelines or Consensuses. First FDA Phase II clinical trial completed Chinese proprietary therapeutic drug for hepatopathy. Substantial clinical evidence shows the efficacy of preventing and treating hepatic fibrosis, liver cirrhosis.

Utidelone Injection is a tumor chemotherapy drug and a Class I innovative drug developed by Huahai Zhongtian (China's first publicly listed synthetic biology pharmaceutical company). (First approved indication) It is indicated for the treatment of recurrent or metastatic advanced breast cancer. Compared to taxane drugs, it offers superior safety, enhanced efficacy, reduced susceptibility to drug resistance, and the ability to cross the blood-brain barrier.

ZAP-X AI radiosurgery robot is dedicated to intracranial tumors and lesions and was specifically developed by the world's leading neurosurgeons, John R. Adler. Through end-to-end AI empowerment, this device establishes a closed-loop system covering from intelligent radiotherapy plans to precise performance. It delivers an innovative solution about dual monitoring and validation system for real-time imaging and dose, enabling highly efficient and precise targeting of the treatment area while protecting surrounding tissue. This approach achieves a double improvement in treatment efficiency and quality. Until April 2026, the system has obtained market approval in 26 countries and regions, including China, and has completed over 7,000 clinical treatments.

As a 12-lead home ECG, BAHEAL® Heartpal Home ECG could meet hospital-level standards. It can accurately monitor a wide range of heart problems, including myocardial ischemia and infarction, which could offer patients with myocardial infarction more critical treatment time. This ECG monitor is portable and easy to operate, making it accessible to non-medical personnel and enabling ECG monitoring anytime and anywhere. The device is also equipped with the functions of doctor’s video consultation, which can provide professional clinical advice to our patients through simple click and connection.
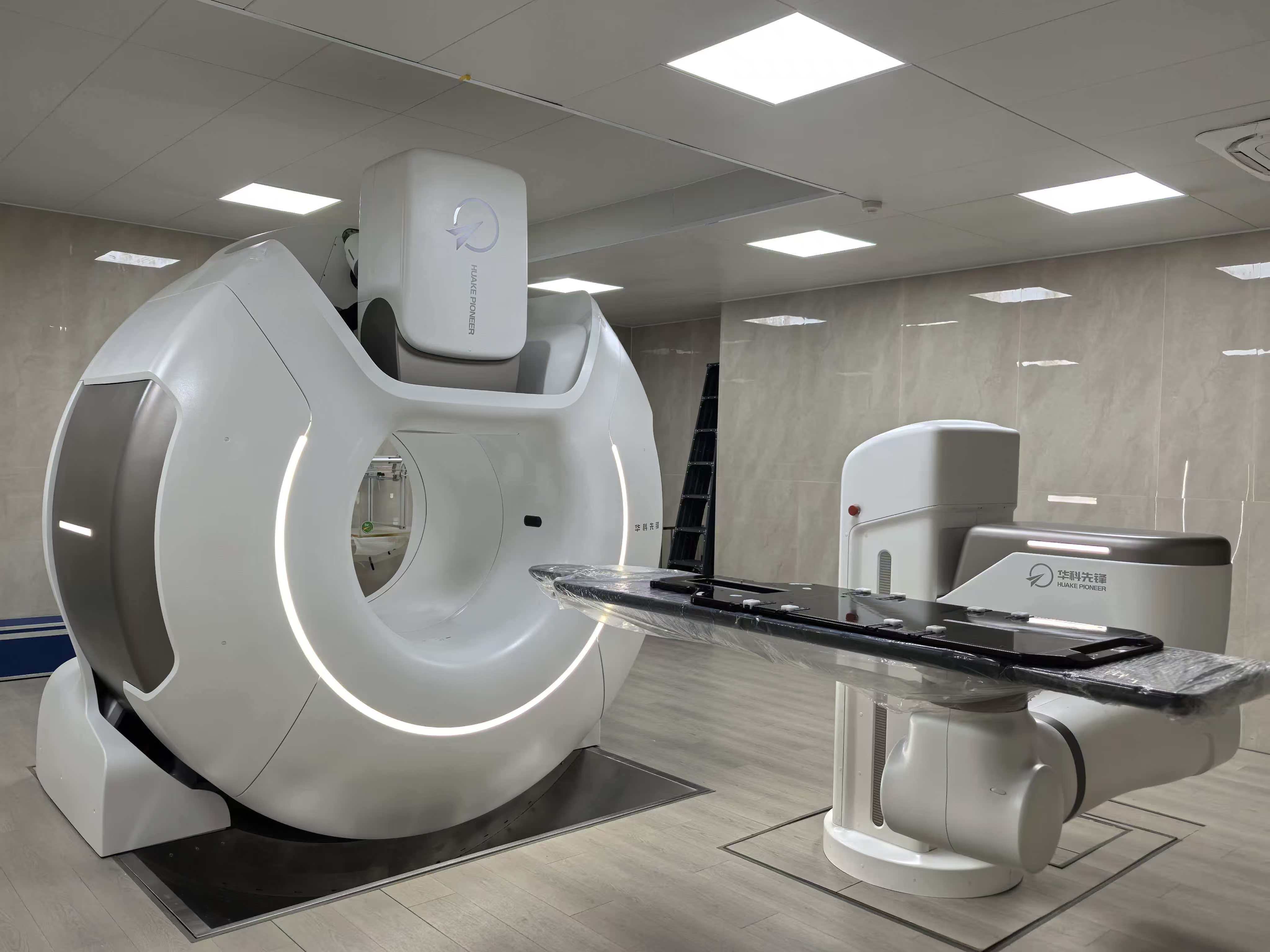
This device is the dual-robot intelligent cooperative radiotherapy system. It’s core component X-band accelerator system was selected into the 13th Five Year Plan Project of the Ministry of Science and Technology in China. At the same time, Huake Pioneer‘s leading heart radiotherapy project was approved for the 14th Five-Year Plan National Key Research and Development Program at the end of 2024.

Cifu® VAD, the fully magnetically levitated ventricular assist device that was developed by BRIOHEALTH TECHNOLOGIES, was officially gained the market approval in China in November 2021. It is China's first and world's only two fully magnetically levitated LVAD, especially in terms of hemocompatibility, implantation invasiveness and operational reliability. Until the end of March 2026, the implantation operations of this product have undergone in more than 100 of China's top cardiovascular hospitals and has benefited more than 800 end-stage heart failure patients.
Besides, as China's first active implantable medical device approved by FDA for clinical trials, BrioVAD® secured Medicare coverage, with each implant covered by $220,000 in Medicare funding. Until now, BrioVAD® has been implanted in more than 200 cases across 26 U.S. clinical centers. Meanwhile, the clinical trials of this product in Europe have been approved in the Netherlands.